
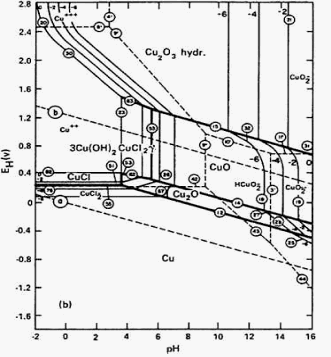
redox equilibria of species not involving hydrogen or hydroxide ions appear as horizontal boundaries (line b).Solid double lines separate species related by redox equilibria (lines c & d).In general, in more dilute solutions, the soluble species have larger predominance areas.reducing the total iron concentration from 1 M to 10 -6 M (more realistic concentrations for geochemists and corrosion engineers) shifts the boundary from pH 1.7 to pH 4.2.reducing the total concentration of Fe 3+ will reduce the driving force of the precipitation.the position of an acid-base equilibrium is dependent on the total concentration of iron.Pourbaix diagrams incorporate Z 1/r calculations and acid-base equilibria.line a shows the pH at which half of the 1 M iron is Fe 3+ and half is precipitated as Fe(OH) 2.Solid lines separate species related by acid-base equilibria (line a).PE values are obtained from reduction potentials by dividing E oby 0.059. The pE scale is intended to represent the concentration of the standard reducing agent (the e -) analogously to the pH scale representing the concentration of standard acid (H +). High E values represent an oxidizing environment. Low E (or pE) values represent a reducing environment. Simplified Pourbaix diagram for 1 M iron solutions. This type of predominance diagram is known as a Pourbaix diagram.E o-pH diagram, or pE-pH diagram. In order to add this variable, we must expand the predominance diagram to include the reduction potential of the environment as well as the pH. You must also take into consideration whether the aqueous environment is well aerated (oxidizing) or polluted with organic wastes (reducing). Knowledge of the pH condition of the environment is not sufficient for predicting the form in which an element will exist in natural waters. However, if suitable reducing agents are present, the phosphorous may not remain in the +5 oxidation state.
The effects of pH on the form in which an element in a given oxidation state exists in natural waters can be summarized with predominance diagrams such as that for phosphorous (V) shown below.
